Clinical Scenario:
In June, we posted the following scenario for comment:

EMS is called to the scene of a “possible stroke”. The patient is a 75 yo female who was last known normal at 8 pm the night before when she went to bed with plans to watch TV before going to sleep. She fell when she tried to get out of bed at 7 am. Her daughter lives with her and heard her fall. When she came into the room, she noted that her mother had a right facial droop, right arm and right leg weakness. She also was unable to speak coherently. The ambulance arrives on scene at 7:30 am and the EMT performs a Cincinnati stroke scale and confirms the findings reported by the patient’s daughter.
The patient lives 20 minutes away from a community hospital which is designated as a primary stroke center. The comprehensive stroke center with endovascular capability is located an hour away.
Where should the patient be taken? What pre-notification alarm bells should be rung? What criteria should EMS systems use to make these transportation decisions in a way that best serves patients without overburdening both the EMS system and comprehensive stroke centers?
We got many thoughtful comments on the above scenario that highlight the complexity of the systems-of-care decisions that face EMS on a local, regional and national level.
Background: The Changing Landscape of Stroke care
Only incremental changes in stroke treatment occurred after the approval of IV tPA in 1996 which established a 3 hour window for IV thrombolysis. After publication of the European trial ECASS III in 2008, the window extended to 3-4.5 hours. But in 2015, a number of clinical trials were published that dramatically increased the management options available for the treatment of stroke patients and challenged the EMS community to change their destination protocols for stroke patients. These 5 trials, MR. CLEAN, ESCAPE, SWIFT-PRIME, REVASCAT, and EXTEND-IA, demonstrated that patients with NIHSS > 6 and proven large vessel occlusion by CT-angiogram may benefit from endovascular reperfusion therapy if they present within 6 hrs of stroke onset [1,2,3,4,5]. A subsequent metanalysis (HERMES) concluded that there may be potential benefit out to 7.3 hours after stroke onset [6].
Similarly to trauma centers, stroke centers are not all equivalent:
– Primary stroke centers (PSCs) provide good stroke care and intravenous tPA
– Thrombectomy-capable PSC can do everything a PSC does, but also has the capability to perform mechanical thrombectomy
– Comprehensive Stroke Centers (CSC) have all of the above, plus extensive resources managing the most complicated patients with dedicated Neuro ICU, neurosurgical services, research and educational resources.
Given the different capabilities of potential destinations, two main issues complicated EMS transport considerations:
(1) To be eligible for endovascular therapy patients had to have a proven large vessel occlusion on CT angiogram. EMS needed to screen effectively for large vessel occlusions in the field using physical exam. A number of scales were developed for this purpose, with varying and less than optimal sensitivity and specificity [7-14, Table 1].
(2) Most of the patients in the above trials received tPA prior to going to endovascular therapy. In many areas, bypassing a primary stroke center capable of administering tPA in favor of going directly to a comprehensive stroke center would place patients out of the tPA window.
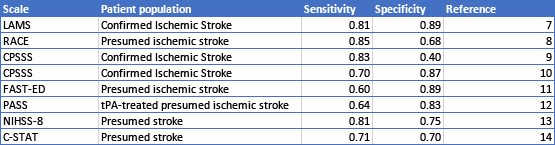
Table 1: Sensitivity and Specificity for Prehospital Screens for Large Vessel Occlusion
Above and beyond this, getting the right patient to the right place at the right time also included considerations for not overburdening Comprehensive stroke centers and excluding primary stroke centers. Primarily in response to the above, the Mission: Lifeline Stroke was formed and developed a Severity-Based Stroke Triage Algorithm for EMS to balance the competing demands of time to tPA, access to endovascular capability and overtriage/undertriage to comprehensive stroke centers.
While EMS was still collaboratively identifying best-practices for patients with possible LVO presenting within 6 hrs of last known normal, two other studies were published last summer which again challenged us reexamine our stroke process of care by extending the time window of patients eligible for endovascular treatment.
The DAWN trial was a prospective, randomized, open-label clinical trial comparing thrombectomy plus standard care vs. standard care [15]. The trial included patients with the following characteristics:
- Last known well between 6 to 24 hrs
- NIHSS > 10
- Imaging-confirmed large vessel occlusion (ICA or proximal MCA)
- Mismatch between severity of clinical deficit and the infarct volume as determined by perfusion imaging.
The trial found a significant difference in functional independence at 90 days (49% for thrombectomy arm, 13% for standard care, p < 0.001) but no difference in 90 day mortality.
The DEFUSE-3 trial was a randomized, open-label trial with blinded outcome assessment that compared thrombectomy + medical therapy vs. medical therapy alone [16]. The trial included patients with:
- Last known well between 6 to 16 hrs
- NIHSS ≥ 6
- Imaging-confirmed large vessel occlusion (ICA or proximal MCA)
- Initial infarct size of < 70 ml
- Evidence of salvageable ischemic tissue on perfusion imaging defined as ratio of the volume of ischemic tissue on perfusion imaging to infarct volume of ≥1.8
Similarly to the DAWN trial, DEFUSE- found a significant improvement in functional independence at 90 days (endovascular therapy 45%, medical therapy 17%, p < 0.001).
Both trials included patients with severe deficits. Mean and Inter-quartile range NIHSS for patients receiving thrombectomy in the DAWN and DEFUSE-3 trials were 17 (13-21) and 16 (10-20), respectively
These trials present several new challenges for EMS transport decisions:
(1) Patients who are clearly ineligible for IV tPA are included, leading to less competition between transport time to comprehensive stroke center (CSC) and tPA eligibility for a significant proportion of stroke patients.
(2) The time criteria are broadened and include the very important population of “wake-up” strokes which made up a significant proportion of the LVO stroke population in the above trials. While broadening the population of patients to be screened for eligibility for endovascular therapy, the criteria are actually very narrow and imaging-based, increasing the possibility of a significant amount of over-triage to comprehensive stroke centers. In one retrospective review of all patients with acute ischemic stroke presenting to a Comprehensive Stroke Center only 1.7% of all patients would have qualified for DAWN enrollment with an additional 0.6 – 1.0% meeting DEFUSE-3 criteria. [17]. Moreover, while CT angiogram may be available at many primary stroke centers, the imaging software (like was utilized for patient selection in both DAWN and DEFUSE III) to evaluate perfusion is unlikely to be.
The comments we received on this post presented a number of responses and potential solutions to these challenges.
Comment Review: The Brainstorming Phase and Regional Solutions
The Right Patient
Dr. Aurora Lybeck made several great comments on this post. She made the very important point that the first step in patient identification starts with the EMT or paramedic at the patient’s side. Stating that “we shall use X… “ to screen for LVO without providing appropriate education and feedback to the provider at the patient’s side will decrease the sensitivity, specificity and utility of validated prehospital LVO screens:
“I think there are a few questions to answer before considering if we SHOULD as EMS to screen for LVO strokes and bypass PSCs for CSCs. 1) Can EMS reliably screen for LVO strokes and 2) What benefit is bypassing PSCs and going straight to a CSC going to have to the patient (a small margin of benefit or a large clinically significant benefit) and 3) what is the acceptable over-triage rates at the CSCs?
With regards to training, we can gather some of the evidence that demonstrates that EMTs can successfully perform one of the screening tests (LAPSS, CPSS, LAMS, RACE, or even a full NIHSS), but the implementation of that in a real-life EMS system with not just new training and competency expectations, but also embedded in a new protocol and transport guidelines that can sometimes be confusing depending on geography etc. If you are one medical director and/or educator and have hundreds of EMTs/paramedics, how are you going to adequately train them all? Have them practice the exam? Ensure competency? Scenarios or simulation? It may be possible with a smaller service or one with robust education but in reality, it\’s an important skill that requires not just skill training but critical thinking and a high degree of clinical competency.” – Aurora Lybeck
If system-design changes are to succeed, they must include plans for involvement and education of the field provider if they are to effectively improve patient outcomes.
The Right Place at the Right Time
From a system standpoint, the outcome benefit of endovascular therapy for a very select group of patients must be balanced with resource utilization within the system as a whole. While it is easy to say that every potential LVO should go to a Comprehensive stroke center, this “transport intervention” could come with a significant amount of unnecessary overtriage that may overburden already-overcrowded centers and add significant cost to the system.
Several of the commenters specifically addressed the issue of over-triage to comprehensive stroke centers. While there was general consensus that embolectomy candidates should be taken to CSC, there was variability in what their path to the stroke center should be. In some cases, it was felt that prehospital LVO scale was sufficient to warrant PSC bypass. In others, there was consideration whether the role of the PSC could still play a critical role in the care of these patients by offering a “secondary screen” in which imaging criteria was used to further narrow embolectomy candidates in such a way that significant time was not lost. While in the end this will vary by region structure and resources, these comments highlight the importance of considering different solutions to the same problem, implementing effective system metrics and measuring patient outcomes:
“If the patient has signs and symptoms of a large vessel occlusion than bypass the primary stroke center for the comprehensive stroke center because tpa alone at the primary may not be effective against the large clot and clot retrieval will be needed anyway, I think???” – Kyle
“RACE LAMS or CPSSS positive for LVO need to go to a comprehensive center. These are the prehospitally validated scales for LVO. If it is to far or time intensive call the helicopter. We are happy to help because time is brain and minutes matter.” – Bill K
“What criteria should EMS systems use to make these transportation decisions in a way that best serves patients without overburdening both the EMS system and comprehensive stroke centers?
– Patient time since last known to be normal
– Willingness of comprehensive stroke center to be OK with a certain amount of over triage.” – Greg Friese
“RACE LAMS or CPSSS positive for LVO need to go to a comprehensive center. These are the prehospitally validated scales for LVO. If it is to far or time intensive call the helicopter. We are happy to help because time is brain and minutes matter.” – Bill K
“We have to be careful when considering this question and come to an answer in a vacuum. EMS triage and destination is critical. But those decisions need to be made in the context of the system of care in region. A regional system of care where the primary stroke center (PSC) can perform a CTA immediately on arrival and upload it to a cloud based imaging viewer that the comprehensive stroke center (CSC) can also immediately review allows the PSC to perform the critical initial function of identifying those that are candidates for embolectomy. Add that to a system where inter-facility transport can be rapidly secured or even auto-launched, the OR can be mobilized ahead of patient arrival, and the patient can be brought directly to the OR at the CSC, and the initial medical contact by EMS to CSC groin puncture time will likely be the same or even less than if the patient was triaged pre-hospital to bypass the PSC and go to the CSC. If too many patients get triaged prehospital to the CSC, then the CSC\’s resources (personnel, scanners, ED beds, neuro beds) may be overwhelmed and their ability to provide care to their LVO-strokes will be compromised. If there is no LVO or they have an LVO, but aren\’t a candidate for embolectomy based on the initial imaging acquired, they can be cared for just as well at the PSC as at the CSC in most cases.” – Chris Zammit
“Great discussion!
In this patient with wake up stroke I would transport to PSC first. Although she does have a LA Motor Score/CSTAT and RACE concerning for LVO she is a wake up and would need both a primary stroke work up (CT to evaluate for hemorrhage and CTA head and neck to identify if she has LVO lesion) It the CTA is positive then perform CTP or DWMRI to evaluate if she fits the criteria set fourth by the DAWN trial or DEFFUSE 3 for reperfusion. I believe most of this can be done at the PSC if there is a prior algorithm with EMS and cooperation for door in door out transfer direct to intervention to the CSC if she has an LVO and fits criteria.” – Rob Dickson
“So, if we decide that based on the evidence, an EMT can indeed be taught the chosen LVO screening exam and can indeed implement it within a new stroke transport destination protocol and can retain the skill and demonstrate competency over time, now how is that implemented in a given service or area? Some protocols will suggest a given time guidelines (ie if there is a CSC within 30 min, or bypassing a PSC would not extend the patient\’s ED arrival more than 20 minutes for example). But there is little to no evidence to guide us on how to geographically on transport- not to mention the reality of time estimates that many of us recognize from practicing in the field. If you plop yourself at any given residential address within your service area, do you know exactly how far you are from the closest PSC? CSC? The difference between those sites? Are they supposed to pull up a map with estimated arrival times and calculate the difference? There is so much subjectivity there, it\’s worth considering all the possible scenarios before implementing a change as important as bypassing a PSC. For some areas, it\’s a moot point. Where I trained in residency and in fellowship, we were in major metropolitan areas, where a CSC is rarely more than 15-20 minutes away. Unless your PSC is in the complete opposite direction and you\’re on the edge of the city, there is likely more to gain and less to lose by choosing the CSC over the PSC- as long as that CSC is willing to accept a lot more stroke patients, knowing that EMS may just default to the “easy” decision of bringing all stroke patients to the CSC (not saying it\’s the right decision, but with complex decision making we know the easiest generalization is often chosen regardless of the protocol minutiae). However, I currently practice in a more rural and suburban area, where the CSC may be over an hour away. For many of our services, it wouldn\’t make any sense to implement screening and new protocols for LVO occlusion when their closest local facility is a PSC and transporting to the CSC would be a delay long enough to exclude some patients from receiving TPA if they are not interventional candidates- those patients are much better served being brought to the local PSC, treated with TPA if eligible, and transported to the CSC for intervention if indeed an LVO and eligible for endovascular intervention- ie the model we are currently using.” – Aurora Lybeck
One other consideration for transport time is patient stability balanced with the clinical skills/training of the field provider:
“Where should the patient be taken?
If I am in the story as an EMT … I am going to the nearest hospital. An hour feels like a long time to be with a patient who potentially needs ALS interventions.” – Greg Friese
“Agree with Greg as well that there are considerations of long transport and risk of airway compromise so provider level of training and capability has to play a role. Also we must consider geographic location and strain on resources from having a truck out of service for 3 hours on this transport.
Lot\’s depends on geography and capabilities of your particular system” – Rob Dickson
In his expert review of this post, Dr. Pete Panagos (Co-Chair of Mission:Lifeline Stroke) wrote the following:
“A big issue also to at least mention is door-in-door-out (DIDO) from PSC to CSC. IF the decision is to always go to nearest/closest stroke center, then the PSC, and EMS, must be committed to rapid identification, evaluation and transfer out, literally within 30-60 minutes of arrival and/or decision to transfer.“
Don’t Forget the Basics…
“Overall, while I think the prospect of identifying LVOs in the field accurately and transporting them to the most definitive care/CSC is exciting and their expedited treatment and recovery is a clinically important outcome to focus on, I don\’t want to lose sight of excellent basic stroke care for all patients. For high functioning urban systems with robust education and training that can implement such new screening skills and protocols, maintain competency, and demonstrate success in patient outcomes and acceptable over-triage rates to the CSCs, I think it\’s great. For most other services though, I think that time in education and emphasis is best spent on excellent basic prehospital stroke care- timely, accurate, checking a glucose and performing a basic Cincinnati stroke scale, appropriate monitoring, sending a stroke alert to the nearest appropriate facility, and bringing the patient straight to CT for the ED team to jump into action. Who knows, maybe someday our more rural services will start identifying LVO strokes and utilizing our HEMS services to get them to a CSC in the future. Thank you to everyone out there putting the time and passion into researching, implementing and closely QA\’ing these new clinical changes. Looking forward to the research that will come out of all the systems out there implementing LVO screening by EMS, and certainly hope to see a significant clinical benefit to patients!
For reference and example, here are some Wisconsin LVO protocols currently in use:
-Milwaukee\’s (using BEFAST): http://county.milwaukee.gov/ImageLibrary/Groups/cntyOEM/EMS/Standards-of-care/Cardio/Stroke2018.pdf
-Madison/Dane County\’s (using FAST-ED, see page 73): https://em-ems.countyofdane.com/documents/pdf/2018%20DRAFT%20EMS%20Protocols%20-%20DRAFT/DCEMS%20Protocols_%203.9.18%20FINAL%20(web).pdf
-LaCrosse/TriState (using FAST-ED, see page 28): http://www.tristateambulance.org/documents/TSA%20Medical%20Guidelines.pdf” – Aurora Lybeck
Last Words
“Why would it be necessary for EMS to make this decision alone? Call stroke alert and report to online medical control” – Mario
Prehospital stroke care does not exist in isolation. The advent of endovascular therapy for stroke challenges the specialty of EMS to take innovative approaches to system design that incorporate best evidence to improve patient outcomes while balancing the strain on resources. The best solutions will consider regional factors, focus on field provider education and value comprehensive quality improvement initiatives that acknowledge the critical role of the EMS provider in the stroke care continuum.
Discussion Summary by EMS MEd Editor, Maia Dorsett MD PhD (@maiadorsett)
Peer Reviewed by Peter Panagos, MD (@panagos_peter)
References:
1. Berkhemer, O. A., Fransen, P. S., Beumer, D., Van Den Berg, L. A., Lingsma, H. F., Yoo, A. J., … & van Walderveen, M. A. (2015). A randomized trial of intraarterial treatment for acute ischemic stroke. New England Journal of Medicine, 372(1), 11-20.
2. Goyal, M., Demchuk, A. M., Menon, B. K., Eesa, M., Rempel, J. L., Thornton, J., … & Dowlatshahi, D. (2015). Randomized assessment of rapid endovascular treatment of ischemic stroke. New England Journal of Medicine, 372(11), 1019-1030.
3. Saver, J. L., Goyal, M., Bonafe, A., Diener, H. C., Levy, E. I., Pereira, V. M., … & Jansen, O. (2015). Stent-retriever thrombectomy after intravenous t-PA vs. t-PA alone in stroke. New England Journal of Medicine, 372(24), 2285-2295.
4. Jovin, T. G., Chamorro, A., Cobo, E., de Miquel, M. A., Molina, C. A., Rovira, A., … & Millán, M. (2015). Thrombectomy within 8 hours after symptom onset in ischemic stroke. New England Journal of Medicine, 372(24), 2296-2306.
5. Campbell, B. C., Mitchell, P. J., Kleinig, T. J., Dewey, H. M., Churilov, L., Yassi, N., … & Wu, T. Y. (2015). Endovascular therapy for ischemic stroke with perfusion-imaging selection. New England Journal of Medicine, 372(11), 1009-1018.
6. Goyal, M., Menon, B. K., Van Zwam, W. H., Dippel, D. W., Mitchell, P. J., Demchuk, A. M., … & Donnan, G. A. (2016). Endovascular thrombectomy after large-vessel ischaemic stroke: a meta-analysis of individual patient data from five randomised trials. The Lancet, 387(10029), 1723-1731.
7. Nazliel, B., Starkman, S., Liebeskind, D. S., Ovbiagele, B., Kim, D., Sanossian, N., … & Duckwiler, G. (2008). A brief prehospital stroke severity scale identifies ischemic stroke patients harboring persisting large arterial occlusions. Stroke, 39(8), 2264-2267.
8. de la Ossa, N. P., Carrera, D., Gorchs, M., Querol, M., Millán, M., Gomis, M., … & Escalada, X. (2014). Design and validation of a prehospital stroke scale to predict large arterial occlusion: the rapid arterial occlusion evaluation scale. Stroke, 45(1), 87-91.
9. Katz, B. S., McMullan, J. T., Sucharew, H., Adeoye, O., & Broderick, J. P. (2015). Design and validation of a prehospital scale to predict stroke severity: Cincinnati Prehospital Stroke Severity Scale. Stroke, STROKEAHA-115.
10. Kummer, B. R., Gialdini, G., Sevush, J. L., Kamel, H., Patsalides, A., & Navi, B. B. (2016). External validation of the cincinnati prehospital stroke severity scale. Journal of Stroke and Cerebrovascular Diseases, 25(5), 1270-1274.
11. Lima, F. O., Silva, G. S., Furie, K. L., Frankel, M. R., Lev, M. H., Camargo, É. C., … & Nogueira, R. G. (2016). Field assessment stroke triage for emergency destination: a simple and accurate prehospital scale to detect large vessel occlusion strokes. Stroke, 47(8), 1997-2002.
12. Hastrup, S., Damgaard, D., Johnsen, S. P., & Andersen, G. (2016). Prehospital acute stroke severity scale to predict large artery occlusion: design and comparison with other scales. Stroke, STROKEAHA-115.
13. Demeestere, J., Garcia-Esperon, C., Lin, L., Bivard, A., Ang, T., Smoll, N. R., … & Parsons, M. (2017). Validation of the National Institutes of Health stroke scale-8 to detect large vessel occlusion in ischemic stroke. Journal of Stroke and Cerebrovascular Diseases, 26(7), 1419-1426.
14. McMullan, J. T., Katz, B., Broderick, J., Schmit, P., Sucharew, H., & Adeoye, O. (2017). Prospective prehospital evaluation of the Cincinnati stroke triage assessment tool. Prehospital Emergency Care, 21(4), 481-488.
15. Nogueira, R. G., Jadhav, A. P., Haussen, D. C., Bonafe, A., Budzik, R. F., Bhuva, P., … & Sila, C. A. (2018). Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. New England Journal of Medicine, 378(1), 11-21.
16. Albers, G. W., Marks, M. P., Kemp, S., Christensen, S., Tsai, J. P., Ortega-Gutierrez, S., … & Sarraj, A. (2018). Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. New England Journal of Medicine, 378(8), 708-718.
17. Jadhav, A. Desai, S., Kenmuir C, Rocha, M, Starr, M, Molyneaux, B, Gross, B, Jankowitz, B, Jovin, T. (2018). Eligibility for Endovascular Trial Enrollment in the 6- to24- hour time window: Analysis of a Single Comprehensive Stroke Center. Stroke. 49:00-00.


